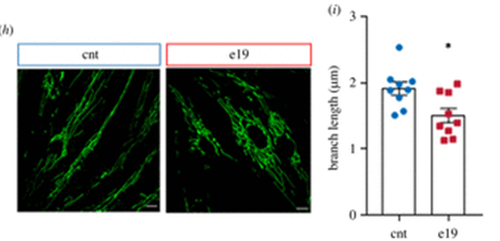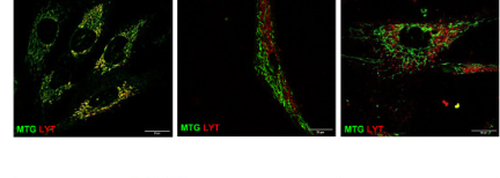
Fig. 1 Assessment of mitophagy by confocal microscopy in control and LIG3 mutant fibroblasts.From left to right, the panels show representative images of control fibroblasts and fibroblasts from patient 1-1 and patient 3-2, co-stained with MitoTracker Green (mitochondria) and LysoTracker Red (lysosomes). Scale bar: 20 μm.
Research themes
Biochemistry of metabolism and bioenergetics studies the cellular chemical processes responsible for energy production and utilization. Metabolism encompasses the reactions that convert nutrients into energy and essential biomolecules, while bioenergetics investigates the mechanisms of energy generation and conservation, particularly mitochondrial oxidative phosphorylation and ATP synthesis. The work of our research group is organized into three main research lines.
1. Study of metabolic and bioenergetic pathways under physiological and pathological conditions
Metabolic and bioenergetic processes are tightly regulated to maintain energy balance and respond to environmental changes. A deeper understanding of these processes is crucial to elucidate the molecular basis of metabolic diseases, aging, neurodegenerative disorders, and cancer. These studies focus on:
- Investigating the role of mitochondrial bioenergetics in regulating cellular homeostasis.
- Analyzing cellular metabolism in diseases such as diabetes, obesity, cancer, and neurodegeneration.
2. Study of the effects of natural bioactive components on metabolism, redox signaling, and cellular bioenergetics
These studies also explore the valorization of food waste. Natural bioactive molecules can modulate key biological processes such as metabolism, bioenergetics, inflammation, and oxidative stress, representing an area of growing interest for medicine and human nutrition. Understanding the molecular bases of their function is important for the prevention and treatment of various diseases. Our research group aims to:
- Analyze the impact of natural compounds on the regulation of carbohydrate, lipid, and protein metabolism.
- Investigate the influence of these substances on ATP production, mitochondrial dynamics, and oxidative stress management.